How To Draw A Lewis Dot Diagram
Introduction to Lewis Structures
The only matter smaller than atoms are their subatomic particles; electrons, protons, and neutrons. Not even under a complex microscopic can we view the individual electrons that surround an cantlet'southward nuclei. The Lewis Dot Structure is a visual which represents the outermost shell of electrons, also known as valence electrons, and possible covalent bonds inside an atom or molecule. These valence electrons are negatively charged and are attracted to the positively charged nucleus, fabricated up of neutrons and protons. Continue in mind that in reality electrons are constantly moving around the nucleus and are not rooted in 1 place every bit portrayed in a 2D structure.
A Lewis Dot Structure is fatigued by a series of dots, lines, and atomic symbols and provides a structure for the way that the atom or molecule is arranged. A Lewis Dot Construction can exist fabricated for a unmarried atom, a covalent chemical compound, or a polyatomic ion.
Using the Periodic Table to Draw Lewis Dot Structures
The periodic tabular array has all of the information needed to draw a Lewis dot construction. Each Grouping, or column, is indicated by a roman numeral which represents the number of valence electrons. This is applicative to the entire group. For example, all elements which fall inside the first cavalcade, or Group I, has one (1) valence electron. All elements in Group 2 have two (two) valence electrons, all the fashion upwards to Eight, viii (8) valence electrons. Properties are also consequent across the rows, or periods, of the periodic table. Periods are indicated past a number, one, two, three, etc. which represent the energy level, or vanquish of electrons. The kickoff period, or row, has simply ane energy level that tin can hold a total of two electrons. Period 2, with a second shell, can hold a total of eight (viii) electrons, also known every bit the octet rule. Menses 3 so along tin concord more than eight (8) electrons.
The periodic table besides conveys electronegativity. The most electronegative elements are located in the uppermost correct corner of the period tabular array and decrease in electronegativity every bit you go down the Group or more left of a Period.
Throughout cartoon Lewis dot structures, the periodic tabular array will exist a potent reference betoken when working with electrons, covalent bonding, and polyatomic ions.
Using Lewis Dot Structures to Show Valence Electrons
Lewis dot structures can be drawn to show the valence electrons that surround an cantlet itself. This type of Lewis dot construction is represented past an atomic symbol and a series of dots. See the following examples for how to depict Lewis dot structures for common atoms involved in covalent bonding.
Example 1. Draw the Lewis Dot Structure for the Hydrogen atom.
Since Hydrogen is in Group I information technology has one (ane) valence electron in its shell.

Example 2. Depict the Lewis Dot Structure for the Florine atom.
Since Fluorine is in Catamenia 2, it tin fit a maximum of viii (8) electrons 2nd energy level. Fluorine Group VII, which means information technology has a total of seven (seven) valence electrons around the atom.

Example 3. Depict the Lewis Dot Structure for Oxygen.
Since Oxygen is in Menstruum 2, it can fit a maximum of eight (eight) electrons 2nd free energy level. Oxygen Grouping Half-dozen, which means it has a full of half dozen (6) valence electrons effectually the atom

Instance A. Decide the total number of valence electrons for C
- Carbon is in Group Iv, four valence electrons
- Full # of Valence Electrons in Carbon = iv
Example B. Decide the total number of valence electrons for H2O
- Hydrogen, Group I, has 1 electron x 2 = 2
- Oxygen, Group Half-dozen, has half dozen electrons x one = 6
- Total Valence Electrons in h2o = 8
Example C. Determine the total number of valence electrons for MgBr2
- Magnesium, Group two, has 2 electrons x 1 = 2
- Bromine, Grouping 7, has 7 electrons 10 two = 14
- Full # of Valence Electrons in MgBrtwo = 16
How to Depict a Lewis Dot Structure
Footstep 1. Determine the total number of valence electrons to be depicted in the Lewis diagram.
Example: COtwo Total = 16
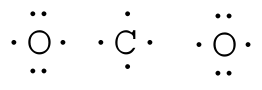
Pace two. Place to the lowest degree electronegative element in heart and draw single bonds from the primal atom to other atoms.
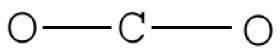
Stride 3. Determine how many electrons must exist added to central element.
Assume that each outer chemical element has a full valence (2 for H, 8 for everything else) from bonding and non-bonding electrons. Total all of these electrons, and decrease that from the total number of valence electrons in the molecule. COtwo has 16 valence electrons. We assume each O has 8 valence electrons. ii×viii=16; xvi-16=0 therefore we don't need to add any electrons to C
Step 4. Add double or triple bonds to central cantlet until information technology has a total octet

Step five. Add electrons to outer elements until they take full octets
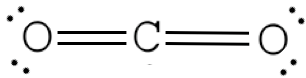
Examples for Cartoon Lewis Dot Structure for Covalent Bonds
Hither, nosotros will be using the determined total number of valence electrons per atom and drawing them in the proper places. Reference the "How to Depict a Lewis Dot Construction" for a Step by Footstep guide. Run across the following Lewis dot construction diagrams for a few covalent compounds.
Example 1. Ammonia, NH3
Nitrogen is in Grouping 5 which means information technology has a total of 5 (five) valence electrons. There are three (iii) hydrogens present, each with their own sole electron giving the unabridged molecule a total of eight (viii) to be accounted for. Since Nitrogen has 5 electrons and is looking for a total of 8 to fulfill its second free energy shell, it is satisfied by the presence of iii hydrogens which fulfills the octet rule. A nonbonding pair of Nitrogen is left and represented a pair of two dots.

Case 2. Methane, CH4
In this structure, there are no nonbonding pairs of electrons present. All have been properly bonded in a serial of lines representing two electrons each. In methane, each Hydrogen has the commencement energy vanquish filled with 2 electrons, its own valence electron forth with a shared electron from Carbon, and Carbon's second energy shell is filled with a full of eight electrons, 4 of its own and 4 shared (ane from each Hydrogen surrounding it).

Cartoon Lewis Dot Structures for Polyatomic Ions
Lewis Dot Structures for cartoon polyatomic ions are done very similarly to that of drawing individual atoms or covalent compounds. However, in this example, we will be dealing with ions rather than simple atoms. Ions are going to possess either a positive or negative charge which should be reflected past the number of electrons drawn as well as an indication of a "-"or "+". This means that there are either additional electrons nowadays to create a negative accuse or less electrons present to create a more positive charge.
Instance 1. POfour iii-, Phosphate ion
With four oxygens present with half-dozen (vi) electrons each and a phosphorus with five (5) there should exist a total of 31 electrons. Nevertheless, since there is a charge of -three.
There are a few things to keep in mind. Phosphorus is in Flow 3, which means it can hold more than 8 electrons and creates a double bail to the oxygen which fulfills the octet rule for one oxygen, but non the others.
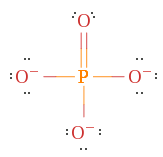
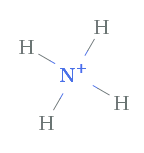
Example 2. NH4 + , Ammonium ion
With ammonium, we are dealing with a positively charged polyatomic ion. The total valence electrons of the nitrogen and iv hydrogens is 9 electrons. Since there is a positive charge of 1+ that means there is 1 less electron, so there will exist a total of viii, which are represented by the four bonds as lines.
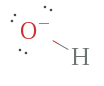
Example 3. OH–, Hydroxide ion
The hydroxide ion has a total of how many electrons? Well, oxygen has 6 and hydrogen has 1, just since at that place is a negative accuse on the ion, it will take an additional ion making a total 8 electrons, which are representing by the bonding pair between oxygen and hydrogen forth with the 3 nonbonding (lone) pairs surrounding oxygen.
Key Concepts:
Determine the total number of valence electrons of the element or chemical compound. If a molecule has more than one element, add the valence electron of all elements present in the compound.
Make up one's mind which atom will exist the central atom of the Lewis Dot Structure. The fundamental cantlet is the to the lowest degree most electronegative atom in the compound. Remember the trend for electronegativity on the periodic tabular array. Once determined, draw that element by atomic symbol in the center and draw unmarried bonds to the other atoms.
Subtract full vanquish of valence electrons (2 for H, eight for everything else) of each outer cantlet from the total number of valence electrons associated with the molecule. Distribute the remaining electrons to the central atom as non-bonding pairs
Form double and triple bonds until the central atom has a total octet.
Describe nonbonding pairs around the outer atoms until they have a full octet.
Bank check your work: Ensure that all of your valence electrons and bonds are accounted for.
Practice with Cartoon Lewis Dot Structures
- Carbon
- Sodium
- Neon
- HCl
- H2O
- And soii
- NO3 –
- ClOthree –
- CN–
- So4 two-
Looking for Chemistry exercise?
Check out our other articles on Chemistry.
Yous tin too find thousands of practice questions on Albert.io. Albert.io lets you customize your learning feel to target practise where you lot need the near aid. We'll give yous challenging practice questions to help y'all achieve mastery in Chemistry.
Commencement practicing hither .
Are yous a teacher or administrator interested in boosting Chemistry student outcomes?
Acquire more than about our school licenses here.
Source: https://www.albert.io/blog/how-to-draw-lewis-structures/
Posted by: emrichhoured52.blogspot.com


0 Response to "How To Draw A Lewis Dot Diagram"
Post a Comment